How to Manage Vaporization in Sampling Systems

How to Manage Vaporization in Sampling Systems
Jon Kestner
It is not easy to vaporize a sample, nor is it always possible. However, if the analyzer in your analytical sampling system requires gas, but the sample is liquid, the only option is to convert the liquid to gas. This process is called vaporization or flash vaporization. The objective is to convert a sample of all liquid to vapor instantly without changing the composition.
If you proceed with this process, it’s important to understand the difference between evaporation--which occurs gradually with an increase in temperature--and vaporization, which occurs instantly with a pressure drop.
It is not possible to vaporize a sample by increasing temperature. Heat causes evaporation, and adding more heat simply makes evaporation happen faster. In a mixed sample, evaporation will allow some compounds to evaporate before others, resulting in fractionation. Vaporization, when done properly, ensures that all of the compounds vaporize at the same time, preserving the sample’s composition.
Things could go wrong when vaporizing. Instead of flashing the whole sample into a vapor, you could unintentionally cause a combination of vaporization and evaporation, resulting in fractionation. Once a sample of mixed compounds fractionates, it is no longer suitable for analysis.
With fractionation, a common scenario is for lighter molecules to evaporate first and travel on toward the analyzer, while the heavier molecules remain behind in the liquid phase. Even if at some later point in the process a fractionated sample appears to be all gas, the mixture will not be of the same molecular proportions as it was before fractionation. It will no longer accurately represent the product taken from the process line. Adjusting process variables--temperature, pressure, and flow – can help ensure proper vaporization and an accurate analytical result.
Understanding the Vaporizing Process
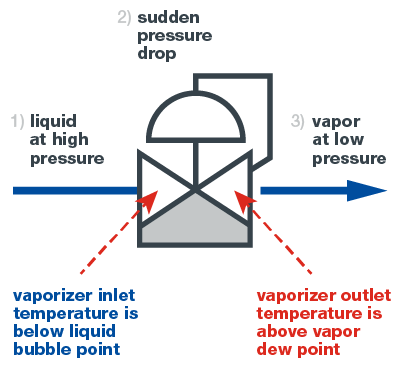
To vaporize a sample, one typically uses a vaporizing regulator, also called a vaporizer. A vaporizer is a pressure-reducing regulator with the capacity to transfer heat to the sample at just the right location. Vaporization consists of a three-stage process, as seen in the diagram below. First, the sample enters the vaporizing regulator as a liquid. At this point, the liquid should not be boiling.
Second, the liquid passes through the regulating cavity in the vaporizing regulator, resulting in a severe and sudden pressure drop vaporizing the liquid. At the same time, heat is applied, enabling the vaporized liquid to remain a vapor.
Third, the sample, now a gas, exits the vaporizing regulator and travels to the analyzer to be read. Due to the immediate transition to the vapor phase, the composition of the gas is unchanged from that of the liquid, ensuring an accurate reading.
In this delicate process, there are many variables or inputs that determine success or failure. For the purpose of this discussion, we will focus on two main sets of inputs.
The first set of inputs relates to the composition of the sample. Depending on its composition, the sample will begin to bubble and will finish vaporizing at different pressures and temperatures. We’ll need to know what these pressures and temperatures are to successfully manage the process.
The second set of inputs concerns settings that you control in your sampling system: pressure, temperature, and flow. Pressure and temperature are controlled at the vaporizer, while flow is controlled downstream at a rotameter (variable area flowmeter) and needle valve. These inputs are set based on what we know about the first set of inputs. Proper vaporization requires a delicate balance of every input. Even when approaching it in a systematic manner like this, the process does require some trial and error.
Understanding the Sample
The best way to understand the first set of inputs is with a phase diagram. A phase diagram plots pressure and temperature, showing at any pair of conditions whether a substance will be vapor, liquid, or solid. The lines indicate the interfaces between two phases.
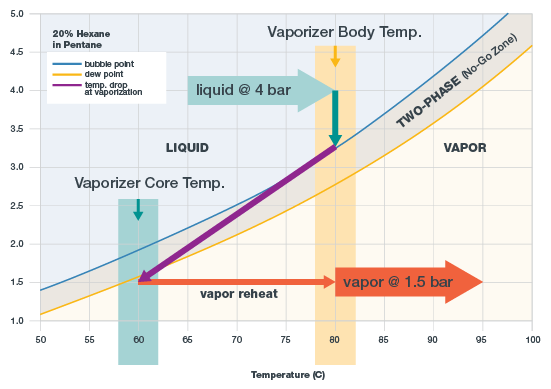
The graph below represents a phase diagram for 20 percent hexane in pentane. When the sample is above the bubble point (blue line), it’s all liquid. The sample should be all liquid when it enters the vaporizer. When the mixture is below the dew point (gold line), it’s all vapor. The sample must be all vapor when it leaves the vaporizer.
Phase diagram showing 20 percent hexane in pentane, with temperature settings
Between the bubble point and dew point lines is the boiling range, or “no-go-zone” of the sample. Here, the mixture is in two phases-- part liquid and part vapor. Once a sample falls into this range, it is fractionated and is no longer suitable for analysis.
With pure and nearly pure samples, there is little, if any, boiling range. The bubble point and dew point lines are nearly superimposed on top of one another. Pure and nearly pure samples will convert to vapor of the same composition, whether through evaporation or vaporization. Some industrial samples approach this level of purity and convert easily.
On the other hand, some samples have such a wide boiling range that they cannot be successfully vaporized. There is no way to pass from the liquid side to the vapor side of the boiling range. The variables—temperature, flow, and pressure—cannot be altered to avoid fractionation.
Most samples fall between these two extremes. For example, in the sample phase diagram, the band between bubble point and dew point is narrow enough so that with the proper settings, the sample can effectively pass from the liquid side to the vapor side of the boiling range. At the same time, the band in is wide enough that the variables must be carefully altered to avoid having a sample in the boiling range.
Setting Temperature, Pressure, and Flow
Converting liquid to gas is a balancing act between the variables. The following is a four-step process for setting inputs for your temperature, pressure, and flow.
- Determine the inlet pressure at the vaporizer. This pressure, which is fixed, is the process pressure, provided the vaporizer is located close to the sample tap. Higher pressure is better because it allows a higher vaporizer temperature, without boiling the incoming liquid.
-
Set the inlet temperature. There are two objectives when setting the temperature. First, the temperature must be low enough that when the sample enters the vaporizer it is entirely a liquid and isn’t bubbling. In the 20 percent hexane in pentane phase diagram, the bubble point at 4 bar is 88°C. To allow a margin of error in maintaining the liquid state, choose 80°C.
The second objective is that the temperature must be high enough to contribute to vaporizing the sample. When you vaporize the sample, the temperature drops, in accordance with the laws of energy conservation. The sample temperature must be high enough at the outset so that after the pressure drop, the sample is not in the boiling range.
- Set the outlet pressure at the vaporizer. The objective is to drop the pressure below the gold dew point line. In the example phase diagram, the outlet pressure is set to 1.5 bar. If the outlet pressure were any higher in this example, the sample would not vaporize entirely and fractionate.
- Set flow. Flow is set downstream at a valve and rotameter, not at the vaporizer. In a sampling system, high vapor flow is desirable because it moves the sample to the analyzer faster. However, high flow can be problematic, too, because more heat is required to vaporize the sample. In other words, high flow results in a greater drop in temperature at the time of vaporization. In the example phase diagram, the purple line illustrates the temperature drop. As flow increases, the purple line angles more sharply to the left.
Another variable influencing the temperature drop is the heat transfer capability of the vaporizer. Some vaporizers are constructed so that heat transfers more efficiently to the sample. When the liquid sample converts to a vapor and its temperature drops, it draws heat from the stainless steel surrounding it. The critical question is how efficiently the vaporizer can replace that heat and keep it flowing to the sample. The more heat the sample can draw, the less its temperature drops during vaporization.
In some instances, it is possible for the vaporizer to be hot to the touch on the outside but cold at the core inside. That’s because the vaporized sample is drawing lots of heat and the vaporizer cannot transfer enough heat to keep up. The best solution is to reduce the flow.
Vaporization involves some approximation. Swagelok Field Service engineers can help your team determine the proper vaporizers for your specific sampling system. As a rule of thumb, keep the flow rate as low as possible without causing an unacceptable time delay in the sample’s travel time to the analyzer. It’s better to start with a low flow rate and experiment with increasing it than to start with a higher flow rate.
Troubleshooting
Phase diagrams help to approximate temperature, pressure, and flow settings, but some troubleshooting will still be required. One sure indication of a problem is poor repeatability in analyzer results. Below we bring up two possibilities when the sample is fractionating instead of vaporizing, with problem #1 being the more common:
Problem #1
Only part of the sample is being vaporized. Liquid is passing through the vaporizer and remains in the tubing on the downstream side. Eventually, it evaporates, and draws heat from the surrounding tubing, making the tubing cold to the touch or causing frost or ice to form. Liquid on the downstream side of the vaporizer may pass beyond the area of the regulator and into other components, such as flowmeters and filters, where it can cause considerable damage.
Solution #1
To solve for this issue, the best option would be to reduce the flow rate. Another option would be to lower the vaporizer outlet pressure, if that is possible. A third option would be to increase the heat to the regulator, but with the added risk of reducing vaporizer life and potentially causing problem #2 (see below).
Problem #2
The sample is boiling at the inlet to the vaporizer. It is fractionating before it can be vaporized. An indicator of this problem is that the inlet tube to the vaporizer twitches, sometimes violently, and the measurement values oscillate. Lighter molecules evaporate and produce a large cloud of vapor molecules. Some of these lighter molecules move on to the analyzer, giving an inaccurate analysis. The remainder create a “vapor wall,” which pushes the liquid back into the process. A portion of that vapor wall then cools and condenses. Finally, the liquid sample moves again toward the vaporizer, where the lighter molecules evaporate, starting the cycle all over again. Eventually, the heavier molecules reach the vaporizer and move on toward the analyzer giving an entirely different analysis.
Solution #2
To solve for this issue, lower the vaporizer temperature.
Conclusion
Vaporizing a liquid sample is challenging. In many sampling systems around the world, vaporizers are constantly fractionating samples and sending unrepresentative samples to the analyzer. You can dramatically increase your chances of success by using a phase diagram of your system’s particular mixture of compounds. You can further increase your success rate by understanding what is occurring in the process—specifically, by knowing what the variables are (temperature, pressure, and flow) and understanding their role in influencing the process outcome. With this framework in place, you can reach the right settings, adjusting in accordance with the signs and symptoms you observe.
Related Articles

Steam Heat Exchangers and Tracing Systems: Best Practices for Design Engineers
When selecting heat exchanger and tracing components for steam systems, it’s necessary design engineers understand the system’s operational characteristics and requirements. Review Swagelok’s best practices to enhance your facility’s steam system.

10 Tips to Improve Sampling Systems
Managing an analytical instrumentation operation is no small feat. Receiving consistent results can be a struggle for even the most seasoned engineers. Luckily, there are several simple tips your team can use to improve your sampling system.